Abstract
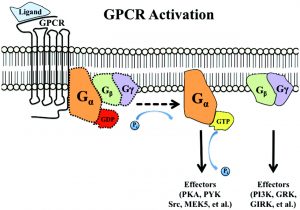
Although several recent studies have reported that GPCRs adopt multiple conformations, it is unclear how subtle conformational changes translate into divergent downstream responses. In this study, we report a new class of FRET-based sensors that can detect ligand/mutagenic stabilization of GPCR conformations that promote G-protein interactions in living cells. These sensors are based on the well-characterized interaction between a GPCR and the C-terminus of a Gα subunit. We use these sensors to elucidate the influence of the highly conserved RY (E/D) motif on GPCR conformation.
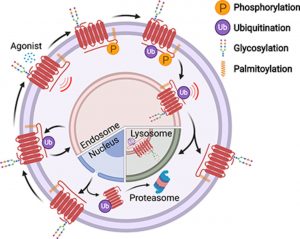
Specifically, Glu/Asp but not Arg mutants of the (E/D)RY motif are known to enhance basal GPCR signalling. Therefore, it is unclear whether ionic interactions formed by the (E/D)RY (ion-locking) motif are necessary to stabilize G-protein coupled Transducer Recombinants (GPCR) basal states. We found that β2-AR(E/D)RY ion-blocking mutagenesis enhances the interaction with Gs. However, only Glu/Asp, but not Arg, mutants increase G-protein activation. In contrast, ion-blocking mutagenesis of opsin (E/D)RY does not alter its interaction with transducin.
Instead, opsin-specific ionic interactions centred at residue Lys-296 are necessary and sufficient to promote interactions with transducin. Efficient suppression of basal β2-AR activity by the inverse agonist ICI 118,551 requires ionic interactions formed by the (E/D)RY motif. In contrast, the inverse agonist metoprolol suppresses interactions with Gs and promotes Gi binding, with concomitant pertussis toxin-sensitive inhibition of adenylyl cyclase activity. Taken together, these studies validate the use of the new FRET sensors while revealing distinct structural mechanisms for ligand-dependent GPCR function.
Keywords: Motive (E/D)RY; 7-Helix receptor; Phone signal; To worry; Functional Selectivity; G protein-coupled receptors (GPCRs); G proteins.
Cell and Calcium Log
HEK293 cells were recently obtained from ATCC (catalogue no. CRL-1573; Middlesex, UK) for reproducibility with GPCR, G-protein, and GAP expression data from the previous studies16. Cells were grown in DMEM (Thermo Fisher Scientific, Waltham, MA) supplemented with 10% FCS and penicillin-streptomycin at 37°C and 5% CO2.
Trypsinized cells were seeded on poly-L-lysine-coated coverslips for 24 min and then loaded with 5 µM Fura-2 AM (Biotium, Fremont, CA) in DPBS without calcium and magnesium (Thermo Fisher Scientific) for 30 min, followed by a wash in HBSS supplemented with 10 mM HEPES pH 7.4 (HBSS-HEPES; Thermo Fisher Scientific). Cells were mounted in a perfusion chamber (Warner Instruments, Hamden, CT) and recorded (excitation (100 ms): 340, 380 nm, emission 520 nm) at one frame per second on a VisiScope Cell Explorer System (Visitor Systems, Puchheim, Germany) equipped with a Plan Neofluar ×40/1.3 oil objective on an Axio Observer.A1 microscope (Zeiss, Jena, Germany), a CoolSNAP HQ2 CCD camera (Photometrics, Tucson, AZ), a VisiChrome polychromator with a 75-watt xenon lamp, and MetaFluor Fluorescence Ratio Imaging software (Molecular Devices, Sunnyvale, CA).
Solutions were changed with the flow controller (Cell MicroControls, Norfolk, VA) and Solution Changer Manifold MSC-200 (Bio-Logic, Seyssinet-Pariset, France), and cells were stimulated with seven different acetylcholine (Ach; Sigma-Aldrich, St. Louis, MO) concentrations in HBSS-HEPES: 100nM, 250nM, 500nM, 750nM, 1.5μM, 3μM, and 10μM. This set of concentrations was selected empirically. It allows detecting a minimum observable cellular response at lower concentrations and reaching a maximum cellular response plateau for higher concentrations.
Ten-second stimulations with each concentration of Ach were repeated five times with an interval of 110 s in which the cells were rinsed with HBSS-HEPES. This temporal protocol was selected taking into account the kinetics of the cell’s Ach response: with the experimental setup used, it takes between 7 and 9 s for a cell to reach the maximum value of the calcium response. Ionomycin (2 μM; Sigma-Aldrich) was added at the end of some experiments.

